SKKU selected in MFDS 'Regulatory Science Talent Development' to establish 'Biohealth Regulatory Science' department
- 약학대학
- Hit3019
- 2021-10-18
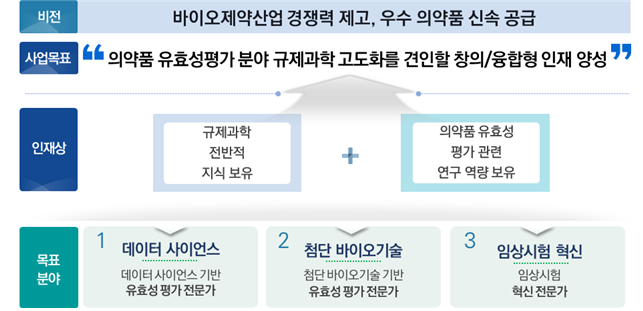
Sungkyunkwan University was finally selected in “University Project for Talent Development in Drug Efficacy Evaluation”, supported by the Ministry of Food and Drug Safety (Research Director, Professor Ju-young Shin, School of Pharmacy). Based on this project, SKKU established the “Department of Biohealth Regulatory Science” in the general graduate school and will receive a total of 2.5 billion won in project expenses for 5 years to foster key master’s and doctoral talents who will lead the advancement of the drug efficacy evaluation field.
The project will be operated based on a cooperative system. A total of 28 faculty members participated in the project, including not only the School of Pharmacy, but also the School of Medicine, Samsung Convergence Medical Science Institute, Samsung Seoul Hospital, Kangbuk Samsung Hospital, Artificial Intelligence College, and pharmaceutical/bio venture companies. In addition, development and operation of a customized curriculum specialized in the field of drug efficacy evaluation, discovery and implementation of projects tailored to demand, student support and field training will be provided.
The Bio-Health Regulatory Science major track is largely composed of “Data Science,” “Advanced Biotechnology,” and “Clinical Trial Innovation”. developed. In addition, in order to strengthen research capacity, we plan to discover and conduct practical-solving research topics needed by industry and government, and we plan to utilize the R&D centers established in the university, such as the Global New Drug Research Center and the Clinical Medicine Research Center, as a research platform. The main participating professors for each track are as follows.
▲Data Science Track: Ju-Young Shin (Pharmaceutical Epidemiology), Ji-Hyeong Lee (Artificial Intelligence), Yoo-Soo Jang (Clinical Epidemiology), Hoon Kim (Computational Biomedicine)
▲Advanced biotechnology track: Choon-Gon Chang (Neuropharmacology), Chung Sang-Jeon (Chemical Biology), Jae-Cheol Lee (Drug Immunology), Jae-Wook Ko (Director of Smart Healthcare Research Center, Samsung Seoul Hospital, Clinical Pharmacology), Hye-Ryeon Chang (Clinical Medicine, Nephrology)
▲ Clinical trial innovation track: Woo-Sung Ho (Director of Clinical Medicine Research Center, Samsung Medical Center, Department of Nephrology), Yeon-Hee Park (Director, Department of Hematology and Oncology, Samsung Medical Center), Joo-Hee Jo (Samsung Convergence Medical Science Institute, Clinical Epidemiology), Jae-Hyeon Lee (Regulatory Science) ), Byungchang Jeong (Director, Laboratory Animal Research Center, Samsung Seoul Hospital, Department of Urology)