Professor Kim Jae-hoon Research Team of the Mechanical Engineering Identify Low-cost carbon battery electrode material
- 공과대학
- Hit5011
- 2020-06-26
Professor Kim Jae-hoon of the Department of Mechanical Engineering, a research team,
Low-cost carbon battery electrode material for high-capacity power storage
ion storage mechanism identification
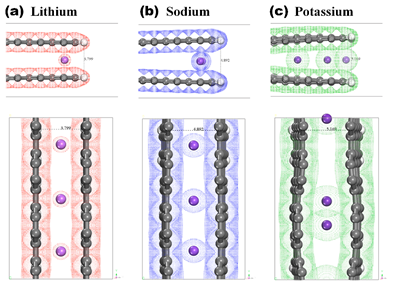
- Identifying lithium, sodium and potassium ion storage mechanisms for hard carbon cathode materials
- Expect new and renewable energy power storage through improved battery stability and capacity
- Advanced Energy Materials, a world-renowned journal, publishes papers and selects journal covers.

The research team led by Professor Kim Jae-hoon of the Department of Mechanical Engineering (first author Stevenus Alvin and researcher Handi Setiadi Cahyadi) announced in collaboration with Professor Kwak Sang-kyu of the Ulsan Institute of Science and Technology that they have identified lithium, sodium and potassium ion storage mechanisms of hard carbon, which have been spotlighted as cathode materials for batteries for medium and large-sized power storage, and presented a new way to develop safe and high-capacity materials.
In order to continuously utilize renewable energy, which does not have uniform electrical power characteristics such as solar and wind power, it is essential to develop a medium and large energy storage system that can store renewable electricity and use it when necessary. However, lithium-ion batteries are having a hard time expanding the base of energy storage systems due to their instability and high. As a result, batteries that store very abundant and low-priced sodium and potassium in hardcabons are in the spotlight, but the ion storage mechanism has not been established, making it difficult to develop high-capacity batteries.
Thus, the research team synthesized the hardcabon as the raw material of rignin and conducted systematic research on the changes in the physical properties of the hardcabon that change during the charging and discharging of lithium, sodium and potassium. Based on the results of the calculation of the density pan-function theory, we also identified that the graphene layer was expanded when previously unknown sodium and potassium were inserted into the hard carbon, and that the graphene layer showed more capacity in the storage of sodium and potassium than lithium, which has no extended effect.
Furthermore, the research team identified storage mechanisms when lithium, sodium and potassium are charged and discharged in hard carbon, and suggested factors for improving capacity and safety of hard carbon cathode materials.
This study is expected to provide measures for improving the safety and capacity of medium and large-sized power storage batteries, and play an important role in the development of new and renewable energy power storage systems.
"By revealing the ion storage mechanism of the hardcabon, which has been controversial, through experiments and theoretical techniques, it can be used to develop low-cost cathode materials for medium and large-sized power storage in the future," said researcher Handi Setiadi Cahyadi.
This study was conducted with the support of the Korea Research Foundation's Climate Change Response Project (2017M1A2A2087635) and the Environmental Industry Technology Institute's Waste Resources Energy Technology Development Project (2018001580001).
The findings were published online on April 15 (Wednesday) in the Advanced Energy Material, a world-renowned journal in the field of materials science, and were published on May 26 (Tue) as a cover for the journal.
※ Name of paper: Intercalation Mechanisms: Revealing the Intercalation Mechanisms of Litium, Sodium, and Potassium in Hard Carbon
https://onlinelibrary.wiley.com/doi/abs/10.1002/aenm Source of the thesis: https://onlinelibrary.wiley.com/doi/abs/10.1002/aenm.202000283